Select image to enlarge
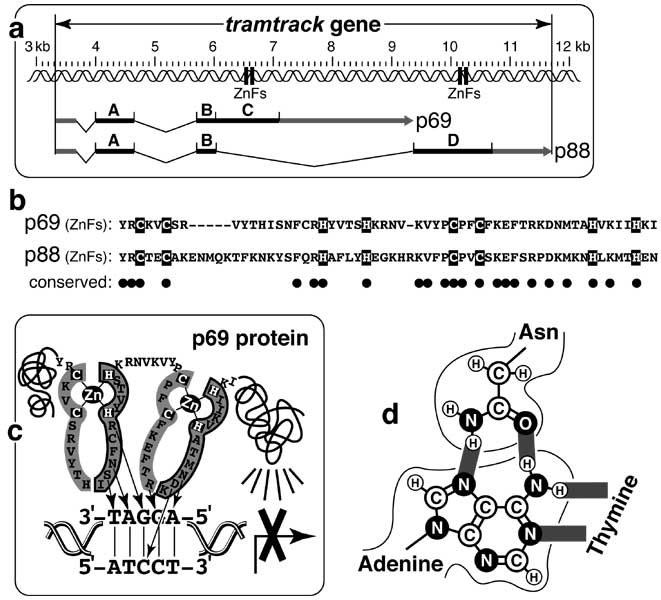
Figure 2.5
Alternative splicing of tramtrack yields different DNA-binding proteins.
a. Transcripts (fragmented arrows) of the ~9 kb gene tramtrack (ttk, 100D) encode two proteins, each of which has different zinc fingers ('ZnFs') [3537, 4773]. Kinked lines are spliced-out pieces, and lightly shaded bars are untranslated sequences. The p69 isoform (641 a.a.) has sections A, B, and C. The 88 kD p88 isoform (811 a.a.) has sections A, B, and D. Shared segments (A, B) encode a BTB protein-interaction domain [211, 1516, 4891], and unshared ones (C, D) encode zinc fingers.
b. Amino acid sequences of the fingers. White letters are Cys and His residues that coordinate Zn2+ ions, dashes are gaps to aid alignment, and black circles mark identical residues.
c. Zinc fingers (shaded) of p69 are drawn flat, though each outlined half is actually an α-helix, and the other half forms β-strands. p69 binds DNA as a monomer [1186], inserting the α-helices of its fingers consecutively into DNA's major groove. Arrows indicate a.a.-base contacts. The DNA oligomer used for structural analysis was devised from p69's enigmatic set of binding sites [1186]. p88's fingers (not shown) bind other sequences [3537, 4775] and have not been studied crystallographically. Transcriptional repression (lines radiating to X'd arrow) might be due to a C-terminal domain (BTB is N-terminal). In some unknown way, p69 (but not p88) is converted from a repressor to an activator in eye development, where the two isoforms act differently in different cell types [2391, 2529]. Both isoforms are regulated by proteolysis [1051, 2529, 4249].
d. Hydrogen bonds (shaded) between asparagine and adenine (cf. N-to-A arrows in c) vs. bonds within the A-T base pair (at right) revealed by crystallography. Such interlocks have fueled hopes for a 'finger code' that could allow fingers to be designed for desired DNA sequences [296, 2265].
This diagram is adapted from [4773] (a), [3537] (b), [1187, 2123] (c), and [296] (d).
|
|
