| Developmental and Signaling Pathways: The Insulin/Tor signaling pathway in fat body and the central nervous system |
| Koyama, T., Mendes, C. C. and Mirth, C. K. (2013). Mechanisms regulating nutrition-dependent developmental plasticity through organ-specific effects in insects. Front Physiol 4: 263. PubMed ID: 24133450 |

|
|
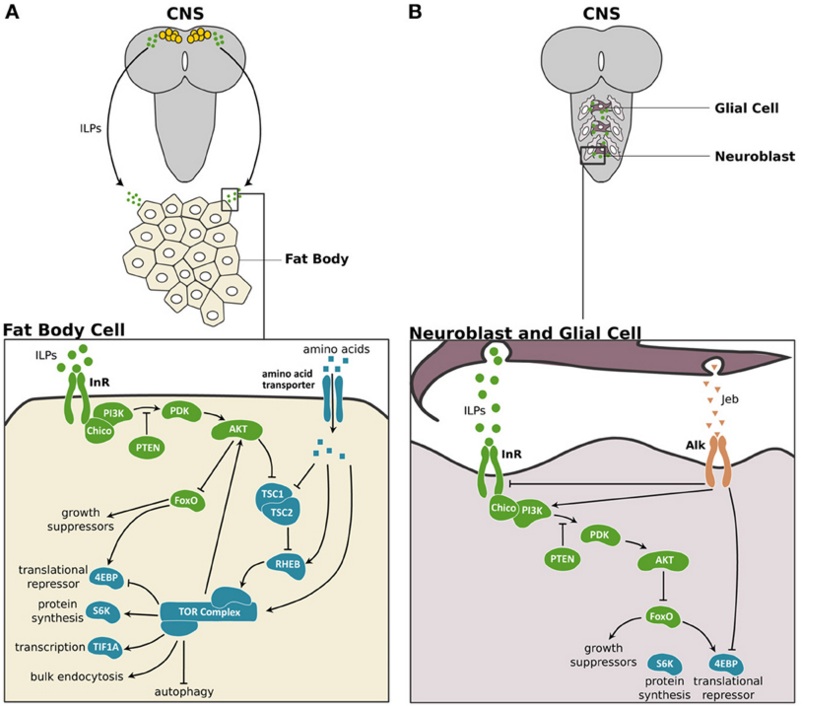
Legend: The Insulin/Tor signaling pathway in fat body and the central nervous system of Drosophila. (A) In Drosophila, three of the eight insulin-related peptides (ILPs) are expressed in a set of neurosecretory cells in the central nervous system (CNS). These ILPs activate insulin/insulin-like growth factor signaling (IIS) in peripheral tissues like the fat body cells. In the fat body, the ILPs bind to and activate the Insulin Receptor (InR), which in turn activates Chico, the insulin receptor substrate. This activation initiates a phosphokinase signal transduction cascade that involves phosphatidylinositide 3-kinase (PI3K), the phosphoinositide-dependent protein kinase 1 (PDK) and the protein kinase Akt. The phosphatase and tensin homolog (PTEN) catalyzes the reverse reaction promoted by PI3K, thereby inhibiting the IIS pathway. When activated, Akt promotes cell growth by inhibiting the transcription factor Forkhead Box class O (FoxO), which activates the 4E-Binding Protein (4EBP), a translational repressor. Furthermore, Akt suppresses the negative regulators of the Target of Rapamycin (TOR) pathway, Tuberous Sclerosis Complex 1 and 2 (TSC1/2). TOR activity itself is enhanced by high concentrations of intracellular amino acids via RHEB. TOR activity promotes cell growth by enhancing translation and ribosome biogenesis through inhibition of the 4EBP and activation of the ribosomal protein S6 kinase (S6K). TOR also stimulates rRNA synthesis by activating the Transcriptional Intermediary Factor 1A (TIF-1A). Lastly, TOR promotes bulk endocytosis and inhibits autophagy. TOR feeds back on the IIS pathway by regulating Akt in a cell autonomous manner. (B) In the CNS two ILPs are secreted by surface glia and activate the IIS pathway in the neuroblasts to regulate the growth of this tissue. The anaplastic lymphoma kinase (Alk) and its ligand, Jelly belly (Jeb) promote growth of neuroblasts in starved larvae. Alk promotes CNS growth in starved conditions by suppressing InR and directly stimulating PI3K activity. IIS components are shown in green, TOR pathway components are shown in blue and Jeb/Alk components are shown in orange. (Koyama, 2013). |
back to Graphic Pathways
Home page: The Interactive Fly © 2016 Thomas B. Brody, Ph.D.
The Interactive Fly resides on the
Society for Developmental Biology's Web server.