In dividing diploid cells the DNA synthetic phase (S phase) is regularly followed by mitosis (M phase). The alternation of G1, S, G2, M and G1 phases is called the cell cycle. In certain circumstances, the M phase does not follow the S phase, and repeated cycles of DNA synthesis results in polyploidy. The process of recurrent duplication cycle without consequent mitosis is called endoreduplication. Instances of polyploid chromosomes in Drosophila include ovary nurse cells, follicle cells surrounding oocytes, abdominal histoblasts (see Escargot), fat body cells, gut cells, and cells of the late prepupal salivary gland. During the process of polyploidization, chromosomes become multistranded. In Drosophila the degree of polyteny is estimated to be approximately 1024 in salivary gland chromosomes, i.e., about 512 in each homolog.
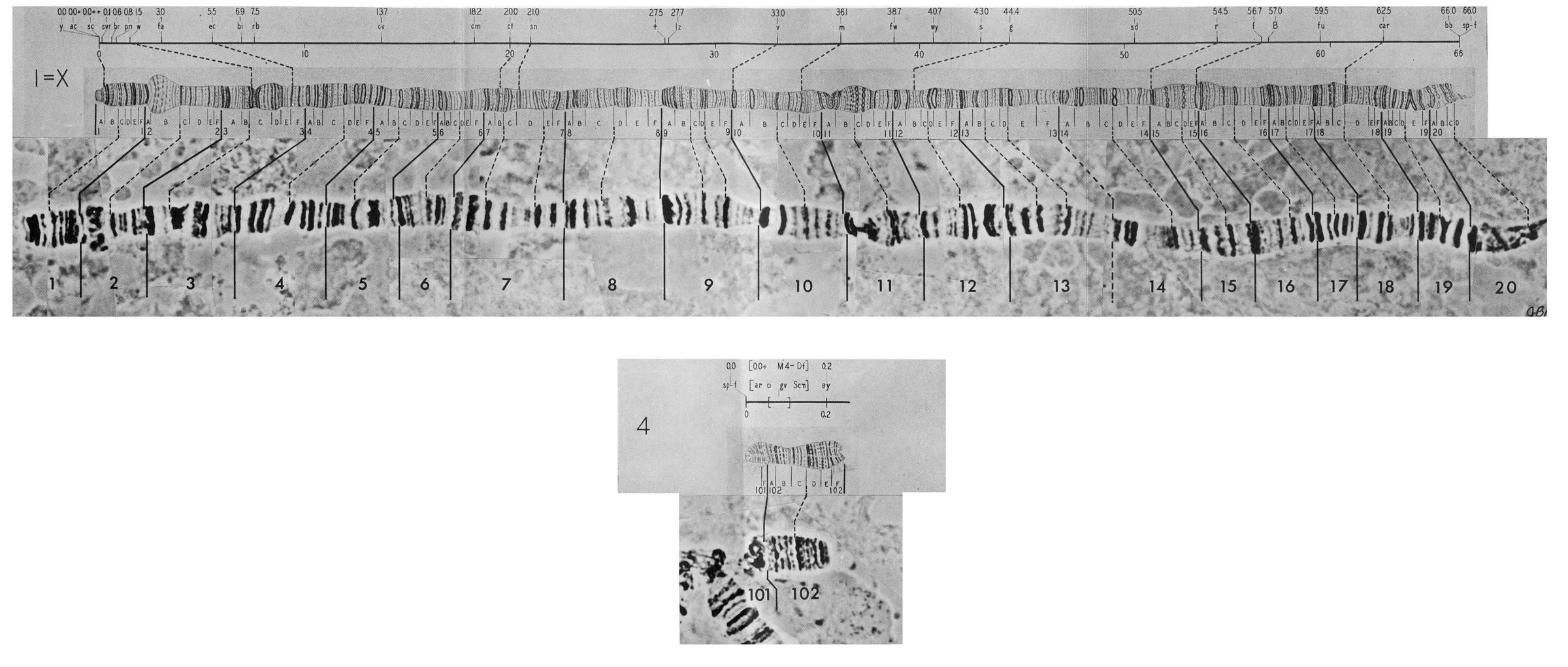
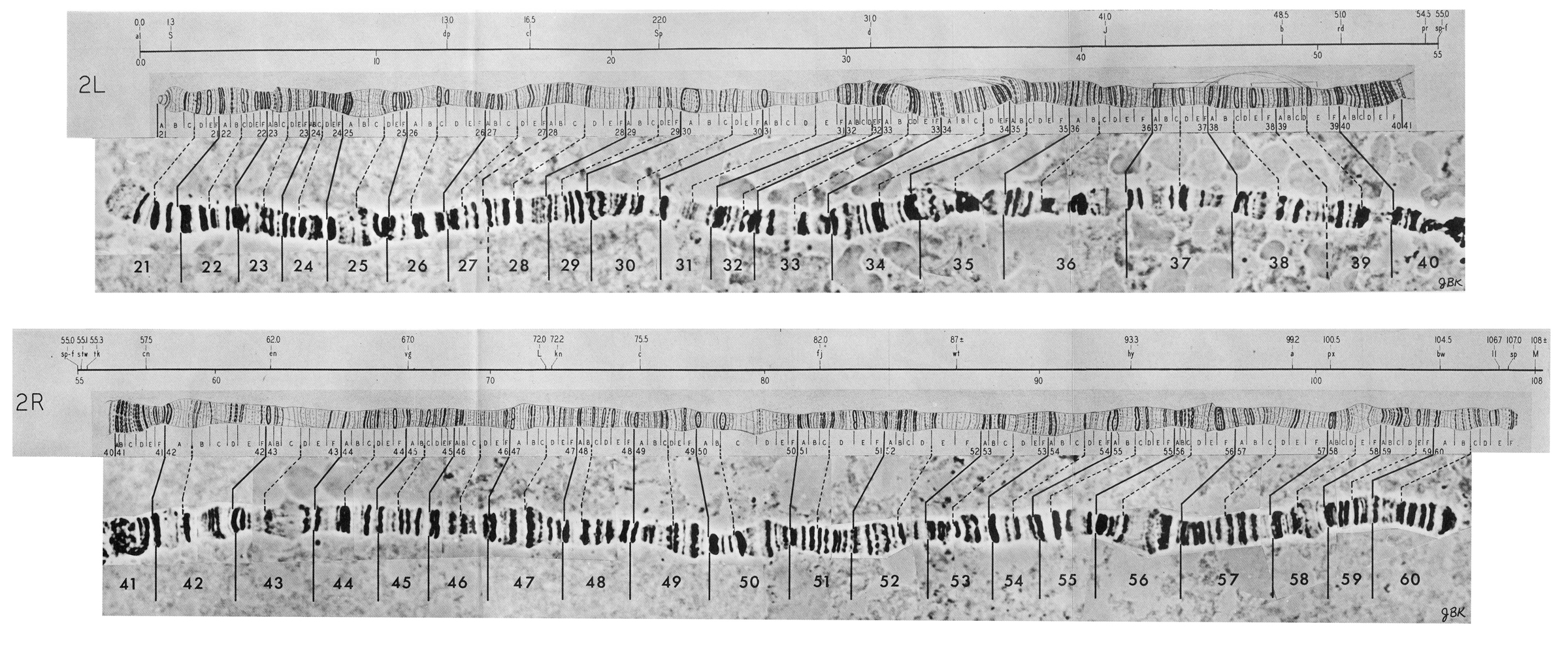
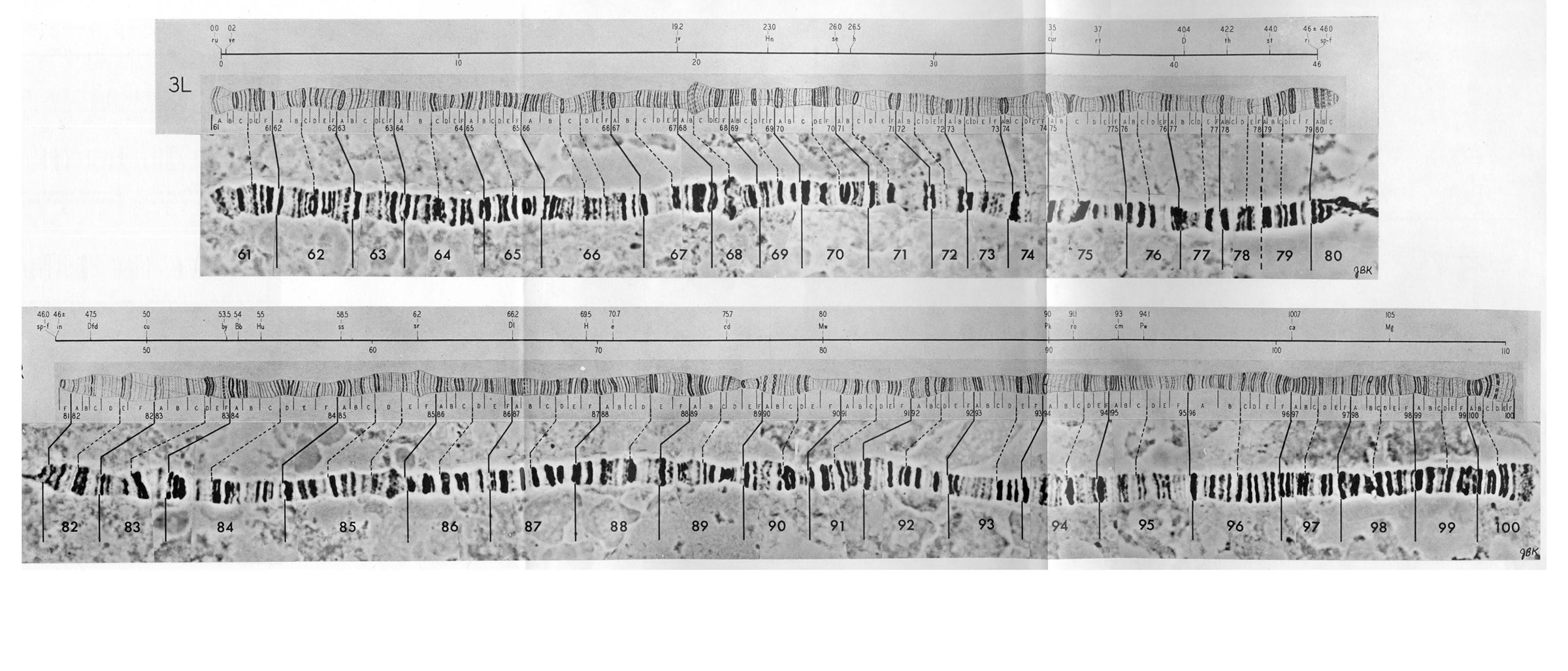
Polyploid chromosomes exhibit a banded structure that is reproducible from individual to individual. In Drosophila there are thousands of recognizable bands. In situ hybridization of cloned complementary DNA of identified genes to banded polyploid chromosomes allows the localization of genes to individual chromosome bands. Chromosomal rearrangements are easily documented by comparing the order of bands between individuals, lines or even species. The degree of rearrangement observed between species is indicative of their evolutionary distance. Drosophila melanogaster has four chromosomes, three autosomes and a pair of sex chromosomes.
The reference system proposed by Bridges divides the limbs of salivary gland chromosomes into 102 sections called "divisions" designated by number from 1 to 102. Each of the five main limbs (X, 2L, 2R, 3L, and 3R) contains 20 divisions; the short chromosome 4 contains only two divisions. The divisions are started with a prominent band and divided further into 6 subdivisions, each designated with capital letters from A to F. Each subdivision starts with a sharp band. Thus each individual band of salivary gland chromosomes can be identified by giving the division number, subdivision, and the number of the band starting from the beginning of the subdivision. Bridges presents the following minimum numbers of bands for the salivary gland chromosomes of Drosophila melanogaster: 537 bands for the X chromosome, 1032 bands for the second chromosome, 1047 bands for the third chromosome, and 34 bands for the fourth chromosome, totalling a minimum of 2650 bands for the whole genome. In this initial count doublets were listed as single bands; more recent interpretations give the total number of bands as 3286 (Sorsa, 1988).
Below:
Polytene chromosomes of Drosophila melanogaster as sketched by Calvin B. Bridges in 1935.
Select thumbnail for larger image.
In late prepupal salivary gland chromosomes, not all DNA in each of the chromosomes is polyploid. Approximately a third of the Drosophila genome is represented by heterochromatin, and heterochromatic regions are underrepresented in polytene chromosomes as these regions do not undergo endoreduplication. For example, the rolled locus is found in a heterochromatic region of chromosome 2 that is considered to remain condensed (and for the most part transcriptionally inactive) throughout all or most of the cell cycle. rolled lies in what is considered to be alpha heterochromatin, a chromosome region that makes up the chromocenter of polytene salivary gland chromosomes. The chromocenter is not thought to be polytenized, that is, it is not thought to undergo repeated rounds of DNA replication resulting in multiple copies of active genes. The chromocenter is thought to be made up of DNA and protein in a dense, tightly knit structure that is transcriptionally inactive. Such heterochomatic regions, which make up 30% of the Drosophila genome, have a much lower density of genes as compared to euchromatin. rolled gene activity is unusual in that it requires the surrounding heterochromatin for gene function. rolled gene activity is severly impaired by bringing rolled close to any euchromatic position; however, these position effects can be reversed by chromosomal rearrangements that bring the rolled gene closer to any block of autosomal or X chromosome heterochromatin (Eberl, 1993).
Puffing is the term that describes structural changes in polytene chromosomes. If one observes polytene chromosomes during the late prepupal stage, different bands appear to be puffed up. For 40 years, this has been understood to be the result of gene activity. Puffs then, afford a view of the temporal sequence of gene activation. A temporal pattern to puffing in the salivary glands of late prepupal flies is inducible by ecdysone injection and is therefore under control of the Ecdysone receptor. A small number of genes react by puffing within minutes of exposure to ecdysone, and a much larger number (>100) react within hours. It is hypothesized that the time sequence of puffing represents a genetic hierarchy of gene activation. Early puffs are independent of protein synthesis while late puffs require prior protein synthesis (Ashburner, 1990). For more information on the hierarchy of gene activation during metamorphosis, see Ecdysone receptor, FTZ-F1 and Broad.
In recent years, transcription factors and chromosomal proteins have been localized to various bands. Binding of these proteins is thought to have functional significance and to reflect the activity of these proteins in gene regulation. For more information on the binding of various proteins and RNA species to bands, see HP1/Su(var)205, Polycomb, Male sex lethal 2, and suppressor of Hairy wing.
An example of binding of specific proteins to polytene chromosomes is found in a study of the protein CHD1 (chromo-ATPase/helicase-DNA-binding domain). Proteins related to CHD1 via the helicase domain have been shown to exist in large multiprotein complexes. For example SNF2/SWI2/Brm proteins (see Brahma and ISWI) are thought to participate in ATP-dependent remodeling of chromatin. Antibodies to CHD1 localize this protein to extended chromatin (interbands) and regions associated with high transcriptional activity (puffs) on polytene chromosomes from salivary glands. These observations support the idea that CHD1 functions to alter chromatin structure in a way that facilitates gene expression (Stokes, 1996).
Polyploidization by endoreduplication requires regulation of the cell cycle. What makes one region of the chromosome become polyploid while another remains underreplicated. Information about the roles of cell cycle genes in the regulation of polyploidization can be found in Cyclin E, Escargot, and Origin recognition complex 2.
References Ashburner, M. (1990). Puffs, genes, and hormones revisited. Cell 61:1-3
Eberl, D. F., Duyf, B. J. and Hilliker, A. J. (1993). The role of heterochromatin in the expression of a heterochromatic
gene, the rolled locus of Drosophila melanogaster. Genetics 134: 277-92
Sorsa, V. (1988). Chromosome maps of Drosophila. Vols. I and II. Boca Raton, Florida: CRC Press
Stokes, D., G., Tartof, K. D. and Perry, R. P. (1996). CHD1 is concentrated in interbands and puffed regions of Drosophila polytene chromsomes. Proc. Natl. Acad. Sci. 93: 7137-42.